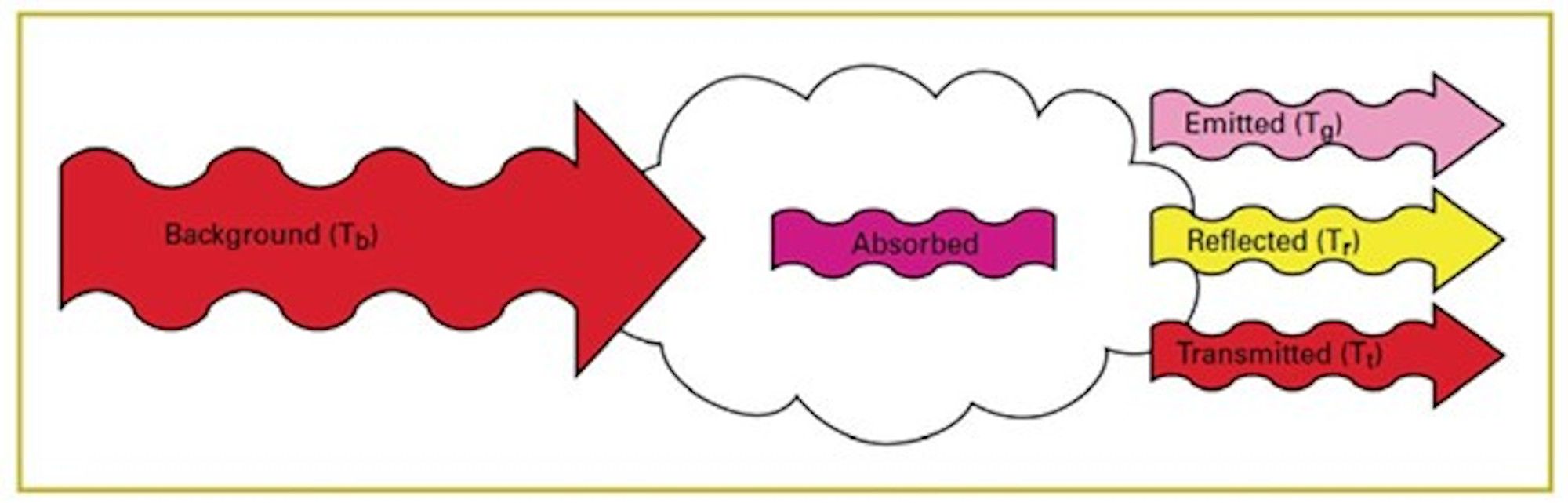
Gas visualisation or Optical Gas Imaging (OGI) is the science of visualising gases. There are various techniques for making a gas visible, but in this article we will discuss gas visualisation by using thermal cameras.
Why can we visualise certain gases in IR?
Thermal imaging cameras measure thermal energy. Due to the rotation and vibration of molecules, certain gases can absorb electromagnetic energy. The energy that can be absorbed, can also be emitted (radiated).
Gases consisting of 1 atom (for example, Helium – He -, Neon – Ne – or Krypton – Kr -) and gases consisting of 2 atoms (Hydrogen gas – H -, Oxygen – O -, Nitrogen – N -) do not create these vibrations. More complex gases like CH4 (methane) or CO2 (carbon dioxide), SF6 ( Sulfur Hexafluoride) are just some of the examples that can absorb electromagnetic energy.
So with a gas detection camera you want to distinguish between the energy absorbed/emitted by the gas itself and the amount of energy coming from the environment.
What is needed to visualise the gas?
A gas detection camera/Optical Gas Imaging camera is built with a sensor or spectral filter that is highly sensitive to the spectral band in which the gas absorbs IR light.
Figure 1: Schematic representation of gas absorption
In order to visualise a gas, there must therefore be a difference in the amount of energy emitted by the gas and its surroundings or background. In practice, the amount of energy reflected by the gas is negligible.
In other words, the greater the difference in radiation between the gas and its surroundings, the better the gas can be detected with the gas detection camera.
Can every thermal imaging camera see gases?
To visualise a gas with a thermal imaging camera, it is important that the gas absorbs light in the spectral band in which the camera is built. Most portable uncooled thermal imagers measure in the 7.5-14µm range.
It is necessary for the thermal imaging camera to be able to detect a radiation difference between the gas and the environment. Most gases are not visible with a standard thermal camera. Water vapour, the gas phase of water, is also not visible in the air to the human eye. As humans, we see the condensation of the gas in the air when the water vapour comes into contact with an air temperature that is below the dew point. Since that water is opaque (not transparent) to a thermal imaging camera, we can see this water vapour better with a thermal camera than with our naked eye.
If a gas has a high transparency of light in the spectral range of the camera, the thermal camera will not be able to detect the gas. These transparency curves as well as the spectral bands of thermal imaging cameras can be consulted online. Thermal Focus can help you with this.

Figure 2: NH3 gas leak diagnosed with FLIR GF306
Cooled versus uncooled gas detection cameras

Figure 3: FLIR GFx320 – VOC Gas Detection Camera

Figure 4: FLIR GF77 – uncooled gas detection camera
Optical gas imagers are among the most sensitive thermal imaging cameras on the market. In order to measure in certain wavelengths at low temperatures, it is necessary to use a sensor that is sensitive in that spectral range.
A distinction can be made between cooled gas detection cameras and uncooled gas detection cameras. When we talk about cooled gas detection cameras, we refer to an IR camera with a sensor that operates at cryogenic temperatures. The sensors are cooled to approximately 70K or -203°C. The most commonly used sensors for the range 3-5µm are the InSb (antimony) detectors. And for the wavelength 8-12µm the QWIP (quantum well infrared photodetector). To cool these sensors a micro stirling cooler is usually used.

Figure 5: FLIR micro stirling cooler
Uncooled gas detection cameras work with a detector that does not need to be intentionally cooled. These types of cameras are also only used in LWIR to visualise gases.
The choice between a cooled and uncooled thermal imaging camera is determined by 2 factors:
- The most important factor is the wavelength in which the camera operates. This wavelength must correspond to the spectral band in which the gas can absorb electromagnetic energy.
- The price/quality ratio
Uncooled sensors are generally cheaper to produce, but have the disadvantage of being able to convert much less IR light into an image, so that the sensitivity is drastically lower than with the cooled thermal imagers. Because they operate in LWIR (8-12µm), the gases visible in the MWIR (3-5µm) part of the IR spectrum cannot be detected at all. The sensitivity of the cooled and uncooled thermal imagers can hardly be compared. Here, the cooled thermal imaging cameras provide superior image quality and sensitivity compared to uncooled cameras, as they suffer less from electronic noise due to cryogenic cooling.
Spectral filtering to visualise gases.
A sensor is built to look through the air with a limited influence of airborne gases such as CO2, H2O, CH4 and O3. These gases reduce the transmission of the IR light through the air. This is why thermal imaging cameras are mainly built in MWIR (3-5µm) and LWIR(8-12µm) and there is little interest in the zone between 5-8µm for example.

Figure 6: Simplified representation of gas detection cameras in the IR spectrum
Although a sensor is sensitive in a wider IR spectrum, a spectral filter is used. The GFx320 is a gas detection camera that has an InSb detector (3-5µm), but has a filter that passes through the spectral band of 3.2-3.4µm. Up to 400 different gases are visible in this spectral band. This is why we also call the GFx320 the VOC camera. VOC stands for volatile organic compounds.
Gases that can thus absorb IR light in the spectrum where the gas detection camera is imaging this part of the spectrum, become visible.
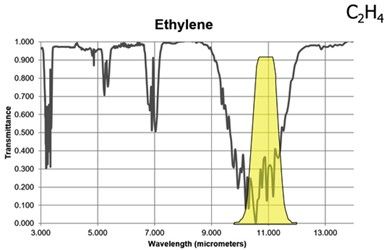
Figure 7: Spectral band of Ethylene compared with the range of FLIR GF306
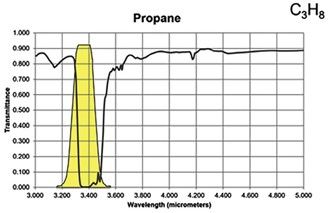
Figure 8: Spectral band of propane compared with FLIR GFx320 and GF620 range
A brief overview of visible gases in IR
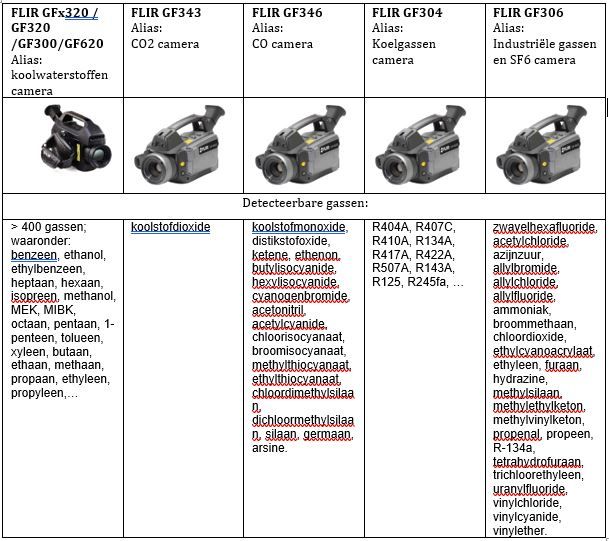

What does a gas leak look like?
In order to visualise a gas, an advanced frame substraction filter is used. In the thermal cameras this can be found as an HSM (high sensitivity mode). The filter is aimed at comparing images over time. There is an interest in a radiation difference that can be detected by the camera. The delta radiation of each pixel is displayed in a smooth manner. Because the gas is in motion, a delta radiation arises due to the concentration differences of the gas.
This creates images like the one below:


Figure 9: CO2 leakage detected with FLIR GF343


Figure 10: SF6 leaks detected with GF306

Figure 11: NH3 Leakage detected with FLIR GF306

Figure 12: CO leak detected with GF346
Figure 13: CH4 leak detected on biogas plant with FLIR GFx320
Andere interessante artikels
What are the differences between cooled and uncooled thermal imaging cameras?
Everyone knows the standard thermal imaging cameras . But did you know that there are also cooled models in our range? These offer a number of interesting advantages.
What is a thermal imaging camera?
A thermal imaging camera is a camera that converts a portion of infrared light from the electromagnetic spectrum (EMS) into an image or video.